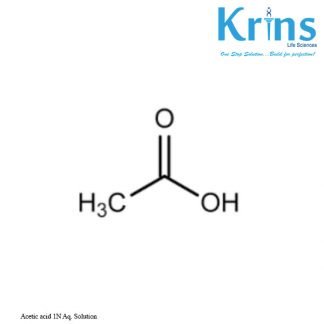
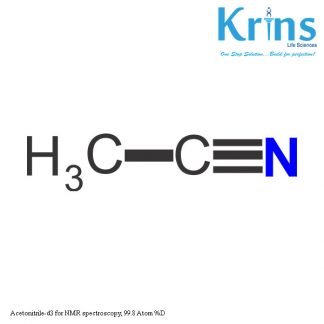
Description
Packing : 500 ml
HSN Code : 38220090
IMDG Identification : Not Regulated for Transport (Non-Haz)
Storage : Room Temperature
Buffer solution pH 9.2
A buffer solution is an aqueous solution containing a mixture of a weak base and its conjugate acid or vice versa.
Buffer Solution pH 9.2 is a clear colourless liquid. The change in pH of Buffer Solution is very little when a little amount of strong acid or base is added to it. Buffer solutions are essential in biology for Calibrating pH meter, maintenance and stability of pH of samples used for testing or preserving the pH of products and test samples for different analytical studies. Buffer is a type of solution which resists any change in the activity of an ion while adding substances that are likely to change the activity of that ion.
Buffer solution is very useful for keeping pH at nearly constant value and there are many systems that use buffering for pH regulation. If the pH of a solution increases or decreases too much, the efficacy of an enzyme decreases in a process. Many biological samples used in research are kept in a buffer solution. The buffering agents are used in chemical analysis and to maintain the stability of various dosage forms. In analytical procedures, it is used to establish hydrogen-ion activity for the calibration of pH meters. It is also used in fermentation processes.
Specifications
| Appearance (Clarity) | Clear |
| Appearance (Colour) | Colourless |
| Appearance (Form) | Liquid |
| pH | 9.2 |